Optimizing Product Formation in Fungal Production Systems
 © AMM / Schmideder
© AMM / Schmideder
Fungal production strains such as Aspergillus niger play a leading role in the large-scale production of fine chemicals, proteins and bioactive agents. However, there is only scant understanding of their regulatory mechanisms as well as the dynamics of cellular processes such as protein transport, secretion, and cell morphology. Yet, these processes significantly limit the efficiency of biotechnological production systems. Applying systems biology and trans-disciplinary approaches makes it possible to reveal and model networks of metabolic, transport and regulations related processes in Aspergillus niger. These then provide the basis for new and optimized paths in genetic engineering, which can be used to overcome the above limitations.
Despite the availability of various fungal expression systems for the production of proteins, enzymes and bulk chemicals, there is still a huge interest within the biotech industry to establish new, superior fungal expression platforms with improved yields. The filamentous fungus Thermothelomyces thermophila is a promising candidate strain, as it naturally secretes lignocellulosic enzymes at high levels. To understand and optimize T. thermophila as a fungal cell factory, we pursue systems biology approaches and develop new synthetic biology tools.
For further information, please contact: Professor Vera Meyer
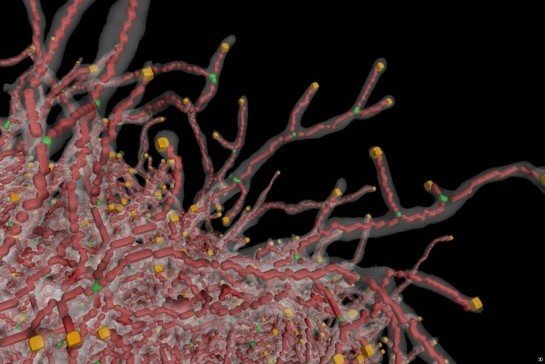
3D recording of Aspergillus niger mycelium using microcomputer tomography:
The recording was made in cooperation with TU Munich’s Chair of Process Systems Engineering and funded by DFG priority program DiSPBiotech 1934.
